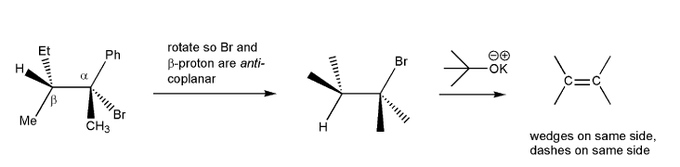
For a molecule to undergo an E2 reaction, the leaving group and the beta-proton must be in an anti-coplanar conformation (one atom straight up, the other straight down). Based on this, which compound undergoes E2 reaction with KOtBu faster? Why?

Using curved hooks, draw a mechanism for the free radical bromination reaction shown below.

Carbocations aren't very stable and so don't last very long after they are formed.
Use curved arrows to show:
a) how a carbocation reacts with a halide ions to form an alkyl halide.
b) how a carbocation reacts with water to form an alcohol.
c) how a carbocation reacts with a base to form an alkene.

Predict the product(s) of the reaction below, and used curved arrows to show a mechanism.
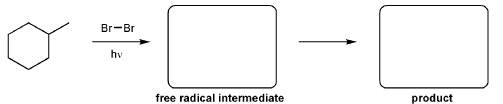
For the reaction below, draw the structures of the radical intermediate and the final product.
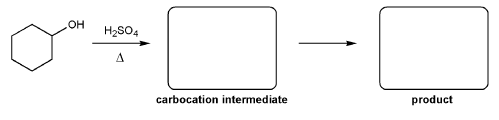
For the reaction below, draw the structures of the carbocation intermediate and the final product.
The alcohol below is protonated and contains an oxygen with a positive charge. Using curved arrows, show the two "legal moves" that result in a neutral oxygen.
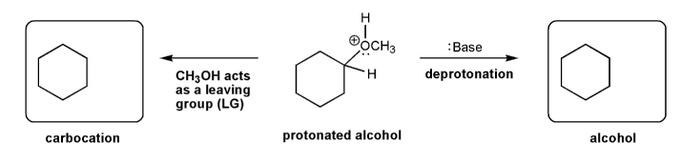
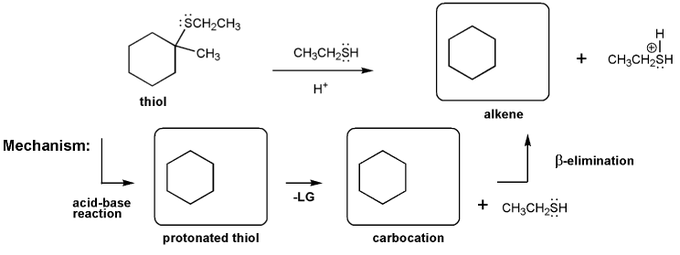
Let's work through an elimination reaction. Draw the structures for each of the species in the three boxes below (protonated thiol, carbocation, and alkene). Also draw curved arrows to show electron movement.
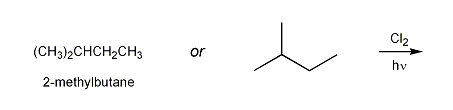
The structure of 2-methylbutane is shown below.
a) Draw the structures of all possible monochloro products resulting from the free-radical chlorination of 2-methylbutane.
b) Based on statistics alone, what do you expect the major product to be? Is this the same structure as the expected major product? Explain.
c) How would the relative yield of the products differ if bromine was used instead of chlorine?