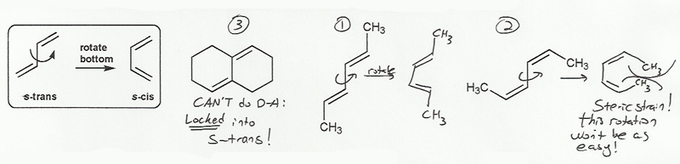
To undergo a Diels-Alder reaction, a dienophile must be in s-cis conformation.
The bicylic compound below is locked into s-trans conformation; it can never rotate into s-cis conformation and so can't undergo a Diels-Alder reaction.
Of the other two compounds, the middle compound most easily rotates into s-cis conformation, and so will undergo a Diels-Alder reaction the fastest. The compound on the right has steric strain when in s-cis conformation, and so won't do Diels-Alder as easily.