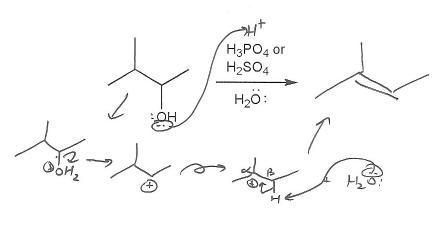
You know this is an elimination reaction because no nucleophile is present; H3PO4 and H2SO4 are non-nucleophilic acids. IF the reagent were HCl or HBr on the other hand, this would be a substitution reaction.
Since acid is present, a carbocation will probably form, so we know that this is an E1 mechanism.
The 2º carbocation that is initially formed will undergo a 1,2-hydride shift to become a 3° carbocation, but that doesn't affect the final product of the reaction; with either the 2° or 3° carbocation, the most stable alkene product is 2-methyl-2-butene.