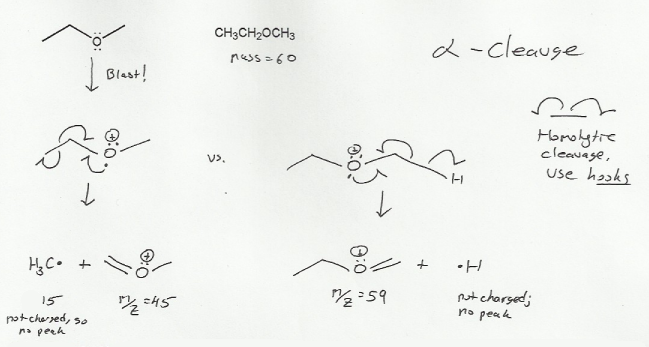
MS ionization will knock off an electron from the heteroatom (atom that's not C or H), in this case, the oxygen, leaving behind a positively charge compound. This is the molecular ion (M+).
Oxygen usually undergoes homolytic cleavage- the bond splits and each atom gets one electron. Since only one electorn is involed, we use hooks instead of the usual curved arrows.
If your professor is into mass spec cleavage mechanisms, I suggest you practice this mechanism, called an alpha cleavage (see image below).